In a thermodynamic system, there are different types of gas laws used. In this post, we are going to discuss different basic gas laws.
Table of Contents
Basic Gas Laws
Different gas laws are
- Boyle’s law
- Charles’ law
- Avogadro’s law
- Dalton’s law of partial pressure
- Ideal gas law
Boyle’s Law
Boyle’s law states that, for a mixed mass of gas at constant temperature volume is inversely proportional to pressure.
Mathematically it can be written as pV=constant
This means if you double the pressure, the volume will get halved.
Explanation: Consider the effect of decreasing the volume of a mixed mass of gas at a constant temperature. The pressure is caused by gas molecules hitting the walls of the container. If the volume of the container is very small, the gas molecules will hit the wall more frequently causing an increase in pressure.
Charles’ Law
Charles’ law states that, for a fixed mass of gas at constant pressure, the volume is directly proportional to temperature.
Mathematically can be written as (V/T)=constant
This means if you double the temperature at constant pressure then the volume will be doubled.
Explanation:
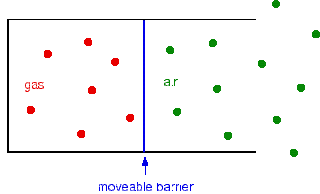
Consider a fixed mass of gas in a container with a movable barrier as shown in fig. The barrier can move without any resistance. The barrier will settle so that the pressure inside and outside is identical.
Now suppose if we heat the gas, but not the air that is outside. The gas molecules will now move faster and will hit the barrier more frequently. On the other side, air molecules are hitting it exactly as before.
It is obvious that the barrier will be forced to the right and the volume of the gas will increase. This will continue until the pressure inside and outside is the same.
So we have fulfilled what Charles’ law says.
Avogadro’s Law
The number of molecules or atoms in a specific volume of ideal gas is independent of the size or the gas molar mass.
Mathematically it can be stated as (V/n)=k; where V is the volume of gas, n is the number of moles of gas and k is a proportionality constant.
Dalton’s Law of Partial Pressure
Dalton’s law or the Law of Partial Pressure states that, the total pressure exerted by a mixture of gases is equal to the sum of partial pressures of the gases in the mixture.
Mathematically it can be expressed asPtotal=Pa+Pb+…………



For latest news you have to go to see the web and on internet I found this web page as a
finest web page for hottest updates.
That is really interesting, You’re a very skilled blogger.
I’ve joined your feed and look forward to in the hunt for extra of your fantastic post.
Additionally, I have shared your web site
in my social networks
Great post. I was checking continuously this blog and I am impressed!
Extremely useful info specially the last part 🙂 I care for such information a lot.
I was seeking this particular info for a very long time.
Thank you and best of luck.
What’s up everybody, here every one is sharing such experience, therefore it’s
pleasant to read this webpage, and I used to go to see
this webpage everyday.
Thank you for the auspicious writeup. It in fact was
a amusement account it. Look advanced to more added agreeable from you!
By the way, how could we communicate?
Appreciate the recommendation. Will try it out.
Today, I went to the beachfront with my children. I found asea shell and gave it to my 4 year old daughter and said “You can hear the ocean if you put this to your ear.” She placedthe shell to her ear and screamed. There was a hermit crabinside and it pinched her ear. She never wants to go back!LoL I know this is totally off topic but I hadto tell someone!